Lengthen Telomeres and Turn Back Aging
Want to live longer and healthier? Of course you do, well science may just have the answer! Scientists have discovered an on-and-off “switch” in cells that may hold the key to healthy aging. This switch points to a way to encourage healthy cells to keep dividing and generating, for example, new lung or liver tissue, even in old age. Getting cells to divide might not be that hard (or even very useful), but that isn’t all, it gets better!
In our bodies, newly divided cells constantly replenish lungs, skin, liver and other organs. However, most human cells cannot divide indefinitely–with each division, a cellular timekeeper at the ends of chromosomes shortens. When this timekeeper, called a telomere, becomes too short, cells can no longer divide, causing organs and tissues to degenerate, as often happens in old age. But there is a way around this countdown: some cells produce an enzyme called telomerase, which rebuilds telomeres and allows cells to divide indefinitely.
In a new study scientists have now discovered that telomerase, even when present, can be turned off.
“Previous studies had suggested that once assembled, telomerase is available whenever it is needed,” says senior author Vicki Lundblad. “We were surprised to discover instead that telomerase has what is in essence an ‘off’ switch, whereby it disassembles.”
Understanding how this “off” switch can be manipulated–thereby slowing down the telomere shortening process–could lead to treatments for diseases of aging (for example, regenerating vital organs later in life).
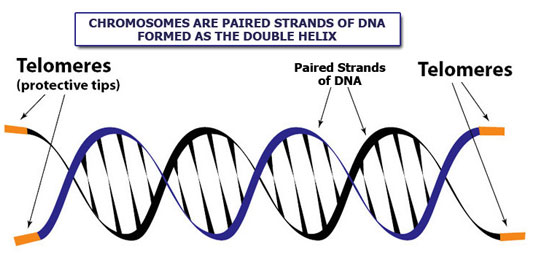
A graphic to show what a telomere is. As you age and your cells divide the telomere tips get shorter (by virtue of they way they get copied). This is thought to be the primary reason we age.
The group conducted their studies in the yeast Saccharomyces cerevisiae, fun fact, it is the same yeast used to make wine and bread. Previously, the group used this simple single-celled organism to reveal numerous insights about telomerase and lay the groundwork for guiding similar findings in human cells.
“We wanted to be able to study each component of the telomerase complex but that turned out to not be a simple task,” Tucey said.
They developed a strategy that allowed the group to observe each component during cell growth and division at very high resolution, leading to an unanticipated set of discoveries into how — and when — this telomere dedicated machine puts itself together.
Every time a cell divides, its entire genome must be duplicated. While this duplication is going on, researchers discovered that telomerase sits poised as a “preassembly” complex, missing a critical molecular subunit. But when the genome has been fully duplicated, the missing subunit joins its companions to form a complete, fully active telomerase complex, at which point telomerase can replenish the ends of eroding chromosomes and ensure robust cell division.
Surprisingly, however, the group showed that immediately after the full telomerase complex has been assembled, it rapidly disassembles to form an inactive “disassembly” complex—essentially flipping the switch into the “off” position. They speculate that this disassembly pathway may provide a means of keeping telomerase at exceptionally low levels inside the cell.
We should keep in mind that although eroding telomeres in normal cells can contribute to the aging process, cancer cells, in contrast, rely on elevated telomerase levels to ensure unregulated cell growth. The “off” switch they discovered may help keep telomerase activity below this threshold and hopefully far from any cancer related activity. It may not help us live forever, but should the research work in humans (a far cry I know) we could, at the least, live healthier and ideally happier.
Sources:
Tucey, T., & Lundblad, V. (2014). Regulated assembly and disassembly of the yeast telomerase quaternary complex Genes & Development DOI: 10.1101/gad.246256.114



But enough about us, what about you?