Day 110: Experiment!
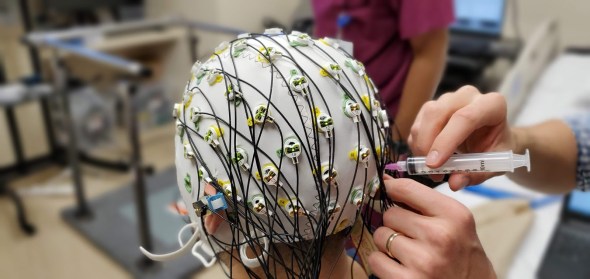
This is an actual photo from my experiment setup, one of my colleagues is adding gel to the sensors. While the giant syringe looks scary, it isn’t. It’s full of conductive gel that gets placed between the head and the sensor. There is no pain or puncturing of the skin involved. Afterwards the gel washes out of the hair. The real pain is sitting for ~30 minutes or so while we gel each electrode enough to get a good reading.
A few days ago I mentioned I did a thing, well an experimental thing really. It was… fun? It was definitely something. Overall it went well, but I said I would give everyone an update and I try to be a man of my word, so let’s do this.
I still feel super hungover from exams. It’s been a super busy week and the experiment that I had to do this week did not make things easier. The good news, it went well and thanks to a colleague in the lab being a data wizard, we have some initial results to show for the confrence next week. Overall it was a success and we learned a lot about what we will be doing when we start the actual experiments.
First, why did we do a dry run? Well, we needed to see how long it would take to do the experiment. Spoiler, it took us approximately three hours. Time wouldn’t be a huge issue if it weren’t for the fact that we have to have our poor volunteer doing something that entire time. So we do a dry run with a lab member to make sure that our protocol is feasible with respect to time, there aren’t any limitations that we may have to deal with (equipment, again time, or even scheduling around our two labs), and lastly we want to make sure that we can collect good data.
When we record noninvasively like this there are some challenges, namely we have to fight to make sure the sensor doesn’t move. Now this is typically an issue for experiments where people are walking, which we are not doing that. However, we are having our subject lay down, so we need to keep the EEG cap (the thing with the sensors) from touching the bed. For our experiment, a towel under the neck seemed to work.
As you can see from the photo and caption, we also have to add a conductive gel between the scalp and the electrode. This gel reduces impedance and gives us a better signal. Like I mentioned it doesn’t hurt, we don’t break the skin, the syringe has a blunt tipped needle and we don’t stab anyone, this is how we add the gel to the sensor, there is a small hole by each sensor that gives us access to add the gel and we place a small amount to close the gap between the sensor and the scalp caused by hair and what not.
Each sensor has a small LED that we can use to visually tell the impedance range for that electrode. Green means we are within working limits, yellow means we are good, but not ideal, and finally red means we need to have a better connection. It’s work to get it all setup, but once it is ready to go, we typically won’t have to mess with it too much so we can set it up and forget it. We do record impedance values before and after the experiment. This can be a good way to validate our recordings as higher impedance will mean lower quality recording.
We also need to make sure that our equipment works well with the equipment from the lab we were working in. I have a fellowship between the school I attend and the clinical lab where I proposed my research. They both have different types of equipment. The EEG cap comes from the school, the rest is from their lab. This is great becuase the clinical lab doesn’t have a super fancy EEG setup like we do (the one shown in the picture) and we don’t have a super fancy synchronization setup like they do in the clinical lab. This means we get the best of both worlds, but also it means we are not guaranteed that our equipment will play nice with there’s.
Now, I cannot show you the results from our dry run, or the experiment itself. Sorry, no fun I know, but on the bright side we will have this published and ready to share well before I finish my year of daily blogging, so you will get an update! I can say that we got some really exciting results even from this initial data analysis and it will be very exciting to start the experiment officially to see what comes out of it.
Until next time, don’t stop learning!



But enough about us, what about you?