Alternatives to antibiotics in an antibiotic resistant world
Let’s be honest, we’ve been getting a little fancy with the antibiotics, creating new and more relevant versions of old favorites like penicillin. Truthfully, we are the problem, how many times do we have to drive home the idea that antibiotics are for bacteria, not viruses. It is not all the consumers fault, the Doctors used to hand out antibiotics to placate angry parents of sick children.
All of which leaves us with a little problem, antibiotic resistance. [1] For those who do not like to click links – or for those who prefer my explanations – antibiotic resistance arises from evolution, or more accurately evolutionary pressure. We are the unfortunate cause of that pressure, by taking antibiotics we are unwittingly selecting for bacteria that are not effected by the antibiotic.
This is important because the lifecycle of bacteria is very short, so while we may never truly see evolution in a human timespan, we can see in in bacteria because they can produce hundreds of generations in a relatively short time. [2] In other words, we humans are the reason for many things; unfortunately, this includes the rise of antibiotic resistance. [3]

So now that we have that covered, the obvious question is what can we do about it? The short answer is, practically nothing. It is just the nature of, well nature, things evolve, and that can have drastic consequences to the very fragile (in comparison) humans that we are.
The long answer is obviously, a little more in depth, there are three semi-feasible solutions available to us right now, all with their benefits and disadvantages. The first would be to keep doing what we are doing, new antibiotics are being discovered on a regular basis (for the moment), and synthetic variants of antibiotics are being tested as well. This of course means that eventually we will run out of options, someday. In a perfect world, this would be enough time for the bacteria to evolve to a point where old antibiotics would work again, however we would need to take them off the market and hope, something I personally would not like to do.

The second option is much more extreme, it would involve finding ways to modify our own cells, in order to block infection. [4] This would be a rather large — but arguably necessary – undertaking especially if humans were ever to travel into space or other worlds, both instances would likely cut us off any source of new antibiotics. Furthermore, if we can get over the fear of the unknown and the advancements in genetic engineering continue at an ever increasing pace, then this solution would not only be viable, we might see it in our lifetime.
That leaves us with the last choice, to that end I want to talk about bacteriophages. For those who are scratching your head right now, bacteriophages (or just phage) are virus’s that attack bacteria. For this reason in the early years after World War 2 when the so-called “iron curtain” was in effect, Russia (then the USSR) spent decades investing heavily in bacteriophage therapy because they did not have access to the antibiotics of the west.
The outcome was good and bad, truthfully because of the cold war the US and other countries shied away from using the technology and to further complicate the problem, the scientific methods of the Russian papers are vague, thus leaving more questions than answers from all the work done. [5]
However, because of antibiotic resistance the push for phage therapy has once again been ignited. To this end, there have been several calls in the US and the UK to further the technology. [6, 7, 8, 9]
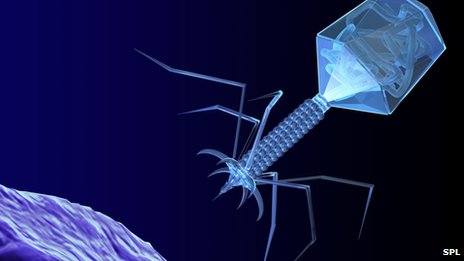
Phage therapy has seen mixed results in the past, given the data collected it seems likely that this is an issue at the genetic level and not with the system itself. Phages are very particular, making them a better candidate to antibiotics – which wipe out good and bad bacteria indiscriminately – however, it could be this very scalpel like precision that is the root cause for the past failures.
However, our generation may succeed where others have failed, gene editing techniques like CRISPR were not around 20 years ago meaning that we could rapidly produce phage therapies for the individual where as before it was almost more of a guess than an actual reason.[10, 11]
Unfortunately, while this would herald the start of truly personalized medicine, it would also mean that phage therapy could not be broad spectrum.
In other words, if you had a bacterial infection made up of bacteria called 1, 2, and 3, then we used phage therapy that treated only 1, 2 or just 3 then you would still have two other strains to contend with. Furthermore, if you were incredibly sick and needed immediate treatment, there would be no time to determine your type of infection and phage therapy would be ineffective at best.
This gap in phage treatment, however temporary, will cement a place for traditional antibiotics for the time being. In addition, phage therapy can still lead to resistance, although from research already done it looks to be about 10 times less than the developed resistance you would see from antibiotics. [5]
While here in the Labs we are hoping to see other options explored, it looks like – in the short term anyway – antibiotics are here to stay and finding new antibiotics are the priority.
However, phage therapy and outright gene therapy are two very promising options that should be funded. Furthermore, if you — our readers — were ever to find yourselves with the opportunity to start research into either of these options, it would not be a chance that we would suggest you pass up.
In the short term, please always finish your antibiotics if you were prescribed them and remember that antibiotics treat bacterial infections, not the flu, not colds, not chicken pox, but bacterial infections.
I hate to end like an advertisement, but only you can prevent antibiotic resistance.

Please, listen to the Batman…
Sources:
[1] WHO (2014). Antimicrobial resistance World Health Organization
[2] Lewis NE, Hixson KK, Conrad TM, Lerman JA, Charusanti P, Polpitiya AD, Adkins JN, Schramm G, Purvine SO, Lopez-Ferrer D, Weitz KK, Eils R, König R, Smith RD, & Palsson BØ (2010). Omic data from evolved E. coli are consistent with computed optimal growth from genome-scale models. Molecular systems biology, 6 PMID: 20664636
[3] Solomon SL, & Oliver KB (2014). Antibiotic resistance threats in the United States: stepping back from the brink. American family physician, 89 (12), 938-41 PMID: 25162160
[4] Tellería-Orriols JJ, García-Salido A, Varillas D, Serrano-González A, & Casado-Flores J (2014). TLR2-TLR4/CD14 polymorphisms and predisposition to severe invasive infections by Neisseria meningitidis and Streptococcus pneumoniae. Medicina intensiva / Sociedad Espanola de Medicina Intensiva y Unidades Coronarias, 38 (6), 356-62 PMID: 24144680
[5] Sulakvelidze, A., Alavidze, Z., & Morris, J. (2001). Bacteriophage Therapy Antimicrobial Agents and Chemotherapy, 45 (3), 649-659 DOI: 10.1128/AAC.45.3.649-659.2001
[6] Reardon, S. (2014). Phage therapy gets revitalized Nature, 510 (7503), 15-16 DOI: 10.1038/510015a
[7] Matsuzaki, S., Uchiyama, J., Takemura-Uchiyama, I., & Daibata, M. (2014). Perspective: The age of the phage Nature, 509 (7498) DOI: 10.1038/509S9a
[8] Ledford, H. (2009). Engineered viruses fight bacteria Nature DOI: 10.1038/news.2009.131
[9] Corie Lok (2001). Antibiotic resistance switched off Nature : 10.1038/news010322-4
[10] Shen, H. (2013). CRISPR technology leaps from lab to industry Nature DOI: 10.1038/nature.2013.14299
[11] Cobb RE, Wang Y, & Zhao H (2014). High-Efficiency Multiplex Genome Editing of Streptomyces Species Using an Engineered CRISPR/Cas System. ACS synthetic biology PMID: 25458909




Your closing thought/message… apt and spot-on. Slap!!
LikeLike
February 1, 2015 at 1:03 pm
Ha thank you!
LikeLike
February 2, 2015 at 1:08 pm
How lucky I am to be married to a genius. xx
LikeLike
February 2, 2015 at 12:51 pm
Well I don’t know about all that, but thank you love.
LikeLike
February 2, 2015 at 1:08 pm