Untangling a cause of memory loss in neurodegenerative diseases

Tauopathies are a group of neurodegenerative disorders, including Alzheimer’s disease that are characterized by the deposition of aggregates of the tau protein inside brain cells. A new study reveals that the cutting of tau by an enzyme called caspase-2 may play a critical role in the disordered brain circuit function that occurs in these diseases.
Use it or lose it: Stopping exercise decreases brain blood flow

We all know that we can quickly lose cardiovascular endurance if we stop exercising for a few weeks, but what impact does the cessation of exercise have on our brains? New research examined cerebral blood flow in healthy, physically fit older adults (ages 50-80 years) before and after a 10-day period during which they stopped all exercise.
Too much activity in certain areas of the brain is bad for memory and attention

Neurons in the brain interact by sending each other chemical messages, so-called neurotransmitters. Gamma-aminobutyric acid (GABA) is the most common inhibitory neurotransmitter, which is important to restrain neural activity, preventing neurons from getting too trigger-happy and from firing too much or responding to irrelevant stimuli.
Embryonic gene Nanog reverses aging in adult stem cells

The fountain of youth may reside in an embryonic stem cell gene named Nanog. In a series of experiments, the gene kicked into action dormant cellular processes that are key to preventing weak bones, clogged arteries and other telltale signs of growing old. The findings also show promise in counteracting premature aging disorders such as Hutchinson-Gilford progeria syndrome.
It’s in the eyes: Alzheimer’s detected before symptoms

Scientists may have overcome a major roadblock in the development of Alzheimer’s therapies by creating a new technology to observe — in the back of the eye — progression of the disease before the onset of symptoms. Clinical trials are to start in July to test the technology in humans.
Alzheimer’s genetics point to new research direction

A analysis of genetic mutations which cause early-onset Alzheimer’s disease suggests a new focus for research into the causes of the disease. Previous research has revolved around the idea that accumulation in the brain of a small, sticky protein fragment — amyloid beta — causes Alzheimer’s disease.
Chemical changes in the brain affect Alzheimer’s disease

A new study is helping to explain why the long-term use of common anticholinergic drugs used to treat conditions like allergies and overactive bladder lead to an increased risk of developing dementia later in life. The findings show that long-term suppression of the neurotransmitter acetylcholine – a target for anticholinergic drugs – results in dementia-like changes in the brain.
Could flies help us understand brain injuries?

Each year, an estimated 1.7 million people in the United States sustain traumatic brain injuries (TBIs), according to the U.S. Centers for Disease Control and Prevention. These injuries occur most frequently from falling, but can also result from military combat, car accidents, contact sports or domestic abuse. Recently, physicians and researchers have become increasingly concerned that even mild cases of repetitive brain trauma could have long-term, unanticipated consequences.
Bad news, fructose alters hundreds of brain genes

Got a sweet tooth? Maybe you even have some sugary goodness with you right now… as you are reading this. Well you may want to put that down.We know a range of diseases — from diabetes to cardiovascular disease, and from Alzheimer’s disease to attention deficit hyperactivity disorder — are linked to changes to genes in the brain. Unfortunately for those who love their pop tarts, a new study has found that hundreds of those genes can be damaged by fructose, a sugar that’s common in the Western diet, in a way that could lead to those diseases.
Limitless: How long-term memories are erased and how to stop it

Currently, neuroscientists think our brain has about enough storage space to hold the entire internet. That’s a lot of space, about a petabyte in fact — if we are to believe this estimate. So, what did you read in the news this day 5 years ago? Don’t worry, I don’t even remember what I had for breakfast this morning and my long-term memory doesn’t fair much better. However, vital information about how the brain erases long-term memories has been uncovered by researchers.
Preventing Alzheimer’s, with an implant

Alzheimer’s has been a losing battle, sure we can fight back with drugs, but that still just prolongs the inevitable. With that said we can all hope this research pans out, to something meaningful. In a cutting-edge treatment for Alzheimer’s disease, EPFL scientists have developed an implantable capsule that can turn the patient’s immune system against the disease. Even better, the implant is subdural, not intracranial.
Alzheimer’s on and now Alzheimer’s off?

Alzheimer’s disease, is anything more frustrating than seeing someone — who otherwise looks healthy — start to forget who you are? Worse than that, we don’t know exactly what causes Alzheimer’s disease, or how to stop it. Well actually that might be changing. Don’t get too excited, because we’ve had false starts before, but an international group of scientists have succeeded in sorting out the mechanism of Alzheimer’s disease development and possibly distinguished its key trigger.
What makes the brain so fast?

Surprisingly complex interactions between neurotransmitter receptors and other key proteins help explain the brain’s ability to process information with lightning speed, according to a new study. Scientists at McGill University, working with collaborators at the universities of Oxford and Liverpool, combined experimental techniques to examine fast-acting protein macromolecules, known as AMPA receptors, which are a major player in brain signaling.
Does your immune system play a larger role in Alzheimer’s disease than thought?

Immune cells that normally help us fight off bacterial and viral infections may play a far greater role in Alzheimer’s disease than originally thought, according to University of California, Irvine neurobiologists with the Sue & Bill Gross Stem Cell Research Center and the Institute for Memory Impairments and Neurological Disorders.
Researchers highlight brain region as ‘ground zero’ of Alzheimer’s disease

A critical but vulnerable region in the brain appears to be the first place affected by late onset Alzheimer’s disease and may be more important for maintaining cognitive function in later life than previously appreciated, according to a new review of the scientific literature.
Certain herpes viruses can infect human neurons

For years, researchers have noted a tantalizing link between some neurologic conditions and certain species of the herpes virus. In patients with Alzheimer’s disease, multiple sclerosis, and cerebellar ataxia, among other neuropathies, the cerebrospinal fluid teems with Epstein-Barr virus (EBV). Yet, the nature of that link has remained unclear, as it has been assumed that EBV, as well as other viruses in the same sub-family, called gammaherpesviruses, cannot infect neurons.
Synapse discovery could lead to new treatments for Alzheimer’s disease
A team of researchers led by UNSW Australia scientists has discovered how connections between brain cells are destroyed in the early stages of Alzheimer’s disease – work that opens up a new avenue for research on possible treatments for the degenerative brain condition.
Scientists to bypass brain damage by re-encoding memories
Researchers at USC and Wake Forest Baptist Medical Center have developed a brain prosthesis that is designed to help individuals suffering from memory loss. The prosthesis, which includes a small array of electrodes implanted into the brain, has performed well in laboratory testing in animals and is currently being evaluated in human patients.
Connecting Alzheimer’s disease and the immune system
The role of the immune system in Alzheimer’s disease is a hot topic, but exactly how the two are connected and what interventions could help lower risk remain a mystery. In a new study, researchers in the Ann Romney Center for Neurologic Diseases at Brigham and Women’s Hospital (BWH) investigate how genetic risk factors for Alzheimer’s disease may influence a key type of immune cell. Their results lay the groundwork for designing better therapeutic strategies and better prediction tools for risk of developing Alzheimer’s disease.
Mexico City’s air pollution has detrimental impact on Alzheimer’s disease gene
A new study by researchers heightens concerns over the detrimental impact of air pollution on hippocampal metabolites as early markers of neurodegeneration in young urbanites carrying an allele 4 of the apolipoprotein E gene (APOE). This is associated with the risk for Alzheimer disease (AD) and a susceptibility marker for poor outcome in traumatic brain injury (TBI) recovery.
A barrier against brain stem cell aging
Neural stem cells generate new neurons throughout life in the mammalian brain. However, with advancing age the potential for regeneration in the brain dramatically declines. Scientists now identified a novel mechanism of how neural stem cells stay relatively free of aging-induced damage. A diffusion barrier regulates the sorting of damaged proteins during cell division.
Cells from human umbilical cord blood improve cognition in Alzheimer’s disease model mice
Alzheimer’s disease (AD), which affects an estimated 26 million people worldwide, is the fourth leading cause of death among the elderly and the leading cause of dementia. Predictions are that the number of AD cases will quadruple by 2050. Although pharmacological methods for treating AD have been discovered, none significantly delay the progression of the disease.
What’s that!? Brain network that controls, redirects attention identified
Researchers at Columbia University Medical Center (CUMC) have found that key parts of the human brain network that give us the power to control and redirect our attention–a core cognitive ability–may be unique to humans. The research suggests that the network may have evolved in response to increasingly complex social cues.
Omega-3 supplements and antioxidants may help with preclinical Alzheimer’s disease
Here’s more evidence that fish oil supplementation and antioxidants might be beneficial for at least some people facing Alzheimer’s disease. A new report describes the findings of a very small study in which people with mild clinical impairment, such as those in the very early stages of the disease, saw clearance of the hallmark amyloid-beta protein and reduced inflammation in neurological tissues. Although the findings involved just 12 patients over the course of 4 to 17 months, the findings suggest further clinical study of this relatively inexpensive and plentiful supplement should be conducted.
DNA breakage underlies both learning, age-related damage
The process that allows our brains to learn and generate new memories also leads to degeneration as we age, according to a new study. The finding could ultimately help researchers develop new approaches to preventing cognitive decline in disorders such as Alzheimer’s disease. Each time we learn something new, our brain cells break their DNA, creating damage that the neurons must immediately repair.
Neuronal disorders and energy metabolism
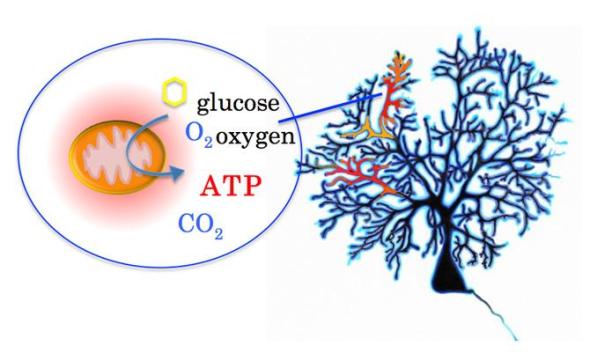
- Mitochondria are responsible for creating more than 90 percent of the energy needed by the body to sustain life and support growth. ATP energy is produced when the mitochondria transfers glucose and oxygen into water and carbon dioxide. How ATP is produced and delivered to intricate neuronal dendrites has been a mystery.
Image credit goes to: Mineko Kengaku, Kyoto University’s Institute for Integrated Cell-Material Sciences (iCeMS)
Scientists in Japan have have discovered how nerve cells adjust to low energy environments during the brain’s growth process. Their study may one day help find treatments for nerve cell damage and neurodegenerative disorders such as Alzheimer’s and Parkinson’s diseases.
Beta secretase inhibitors offer treatment for Alzheimer’s disease
With each new amyloid-targeting treatment for Alzheimer’s disease that has been developed, there has been a corresponding concern. For example, antibodies targeting amyloid-β peptide (Aβ) produce inflammation in the brain in some patients. Gamma secretase inhibitors tend to produce adverse effects by interacting with Notch, an important pathway for cellular signaling. However, a new target for alzheimer’s is offering some new hope.
Immunotherapy, a promising new treatment of Alzheimer’s disease
Alzheimer’s disease, it slowly takes things away from the person without giving anything back. Right now there is no cure and at best we can slow the progression in some cases. Time is always a factor and no two cases are the same. However, new treatments are in the works and a new study has revealed that a single dose of an immunotherapy reverses memory problems in an animal model of Alzheimer’s disease.
Alzheimer’s, the autoimmune disease?
Brain levels of the lipid ceramide are high in Alzheimer’s disease, and now scientists have found increased levels of an antibody to the lipid in their disease model. While some members of this lipid family are a plus in skin cream, inside the brain, ceramide appears to increase beta amyloid production and help the iconic plaque kill brain cells in Alzheimer’s.
Possible mechanism underpinning Alzheimer’s and Parkinson’s type diseases found
Neurodegenerative diseases have remained stubbornly increasing in prevalence for sometime now. Unfortunately longer life does not mean a better quality of life. Thankfully that could change sooner than you think, scientists have for the first time discovered a killing mechanism that could underpin a range of the most intractable neurodegenerative diseases such as Alzheimer’s, Parkinson’s and ALS.
New Protein Implicated in Alzheimer’s
Alzheimer’s prevention has made some strides in recent years. We’ve even identified some new causes, and in some cases we can do both. In fact, researchers have now shown that low levels of the protein progranulin in the brain can increase the formation of amyloid-beta plaques (a hallmark of Alzheimer’s disease). These plaques can cause neuroinflammation, and worsen memory deficits in a mouse model of this condition. Conversely, by using a gene therapy approach to elevate progranulin levels, scientists were able to prevent these abnormalities and block cell death.
New test for Diagnosing Alzheimer’s Early
Alzheimer’s diagnosis is important, like the famous slogan “with a stroke, time lost is brain lost,” detecting alzheimer’s is important in order to stave off cognitive decline. A just like a stroke time lost is brain lost. Unfortunately early diagnosis has been hard to come by, but now researchers say a simple test that combines thinking and movement can help to detect heightened risk for developing Alzheimer’s disease in a person. The best part, they say this will work even before there are any telltale behavioural signs of dementia.
New ways to test for Alzheimers
Accurately diagnosing alzheimer’s is not an easy thing to do. In fact most of the time people aren’t diagnosed until very late in the progression of the disease, long after serious damage to the brain has been done. Biological markers of Alzheimer’s disease may be able to detect it at an earlier stage. For example, using brain PET imaging in conjunction with a specialized chemical that binds to beta-amyloid protein, the buildup of the protein as plaques in the brain can be revealed years before symptoms appear. But as with anything that requires any sort of imaging techniques, these scans can be expensive and are not available everywhere. That is all about to change thanks to four new studies that aim to help offer multiple ways to test for alzheimers much earlier than we can currently test.
Alzheimer’s? Forget about it!

Let’s face it, diseases that affect the brain are some of the hardest to deal with. Anyone who’s lost someone to alzheimer’s knows exactly what I am talking about. Looking at someone you love and watching them slip away without any visible difference can be heart wrenching!
However, there is a new hope! Physicists [of all things] from Michigan State University published an article in the Journal of Biological Chemistry, that might make alzheimers and associated diseases a thing of the past.