The long-term effects of COVID


Sometimes things just work out a particular way. Two days ago I got the bivalent COVID vaccine and that same day Nature Medicine released a new paper on the long-term effects of COVID infection. The paper has already been getting a lot of attention and as you would imagine people are quickly trying to downplay the implications, because the facts are not looking good to those who want to minimize COVID. In fact, at this point ignoring the dangers is ignoring reality and sadly people seem to be okay with pushing that ignorance. So let’s talk COVID… again.
(more…)Everything you should know about the COVID vaccine

I’ve already written several posts on my experience with the first COVID vaccine dose (here and here) and the second dose (here and here). Today we’re putting it all together. I’m going to run through everything you should know about the COVID vaccine from how it works, to why you should get it, and what to expect when you go through the shots. My goal with this is to make it accessible as possible so you can make an informed decision and feel comfortable getting the shot. Sometimes the science is hard to understand, but I think we can simplify it enough to make sense to just about everyone. That said let’s give it… a shot.
(more…)COVID Vaccine: The second shot

Today is the day! It’s been roughly two weeks (if I’m counting that right) since I got my first COVID-19 vaccine shot and today I’m getting my booster, or second shot, whatever we’re calling it. For that reason, today we’re going to recap the last two weeks and discuss what may happen with the second shot. It’s not as exciting as it sounds, but there’s a lot of misinformation out there so it’s important to document all this.
(more…)Day 309: No tests, no worries


If I ignore my bills, I would have very few, if any. If that logic seems, well wrong, then I’m not surprised. Ignoring something doesn’t make it magically go away. Yet, Trump has touted, yet again, that if we slowed or stopped testing, we would have very few, if any cases of COVID19. I mean, with that kind of logic, you could have no more bills with this one weird trick!
The science behind real life zombies

In the spirit of Halloween we bring you the science fact and fiction behind the undead. Zombies, those brain loving little guys, (and girls) are everywhere. Sure, we are all familiar with the classic zombie, but did you know that we aren’t the only zombie lovers out there? It turns out that nature has its own special types of zombies, but this isn’t a science fiction movie, this is science fact! Sometimes fact can be scarier than fiction, so let’s dive in.
Antibody therapy opens door to potential new treatment for HIV

The development of antiretroviral therapy, a combination of drugs that slows the replication of HIV in the body, has transformed the treatment of this infection. What was once a certain death sentence is now a chronic condition that people can live with for decades. But this therapy has drawbacks. There are side effects, including kidney problems, decreased bone density, and gastrointestinal problems. And if a person discontinues his or her treatment, even missing a few doses, the level of the virus in the body is able to rebound quickly.
Study finds vast diversity among bacteriophages

Viruses that infect bacteria are among the most abundant life forms on Earth. Indeed, our oceans, soils and potentially even our bodies would be overrun with bacteria were it not for bacteria-eating viruses, called bacteriophages, that keep the microbial balance of ecological niches in check. Now, a new study suggests that bacteriophages made of RNA — a close chemical cousin of DNA — likely play a much larger role in shaping the bacterial makeup of worldwide habitats than previously recognized.
Ancient viruses lurk in our DNA

Think your DNA is all human? Think again. And a new discovery suggests it’s even less human than scientists previously thought. Nineteen new pieces of non-human DNA — left by viruses that first infected our ancestors hundreds of thousands of years ago — have just been found, lurking between our own genes.
Viruses ‘piggyback’ on hosts success

In the microscopic life that thrives around coral reefs, researchers have discovered an interplay between viruses and microbes that defies conventional wisdom. As the density of microbes rises in an ecosystem, the number of viruses infecting those microbes rises with it. It has generally been assumed that this growing population of viruses, in turn, kills more and more microbes, keeping the microbial population in check. It’s a model known as “kill-the-winner” — the winners being the blooming microbial cells and the killers being the viruses (mostly bacteria-killing viruses known as bacteriophages) that infect them.
Zika virus infects human neural stem cells, but…

The Zika virus infects a type of neural stem cell that gives rise to the brain’s cerebral cortex, Johns Hopkins and Florida State researchers have found. On laboratory dishes, these stem cells were found to be havens for viral reproduction, resulting in cell death and/or disruption of cell growth. While this study does not prove the direct link between Zika and microcephaly, it does pinpoint where the virus may be doing the most damage.
A way to track and stop human and agricultural viruses

Viruses are molecular thieves that take from their hosts under the cloak of darkness. But now a Virginia Tech scientist has found a way to not only track viral hijackers, but also potentially stop them from replicating. The discovery has broad ranging applications in stopping viral outbreaks such as Hepatitis C in humans and a number of viruses in plants and animals because it applies to many viruses in the largest category of viral classes — positive-strand RNA viruses.
The world’s smallest terrorist: Virus hijacks protein machine and then kills the host

Bacteriophages are viruses that infect and kill bacteria. The name originates from the Greek ‘phagos’ which means ‘to devour’. Bacteriophages were discovered 100 years ago because of their ability to replicate in a pathogenic bacterium, kill it and thereby cure the patient. As a small spaceship landing on the moon, the microscopic particles land on the surface of the bacteria where they inject their deadly genetic material.
Seeing viruses in a new light

Want to make a virus? It’s easy: combine one molecule of genomic nucleic acid, either DNA or RNA, and a handful of proteins, shake, and in a fraction of a second you’ll have a fully-formed virus. While that may sound like the worst infomercial ever, in many cases making a virus really is that simple. Viruses such as influenza spread so effectively, and as a result can be so deadly to their hosts, because of their ability to spontaneously self-assemble in large numbers.
Certain herpes viruses can infect human neurons

For years, researchers have noted a tantalizing link between some neurologic conditions and certain species of the herpes virus. In patients with Alzheimer’s disease, multiple sclerosis, and cerebellar ataxia, among other neuropathies, the cerebrospinal fluid teems with Epstein-Barr virus (EBV). Yet, the nature of that link has remained unclear, as it has been assumed that EBV, as well as other viruses in the same sub-family, called gammaherpesviruses, cannot infect neurons.
Closing the loop on an HIV escape mechanism
Nearly 37 million people worldwide are living with HIV. When the virus destroys so many immune cells that the body can’t fight off infection, AIDS will develop. The disease took the lives of more than a million people last year.
A protein-RNA structure hints at how viruses commandeer human proteins
Researchers at Case Western Reserve University and the University of Michigan have produced the first image of an important human protein as it binds with ribonucleic acid (RNA), a discovery that could offer clues to how some viruses, including HIV, control expression of their genetic material. That information could lead to new strategies to block viruses from replicating, thereby limiting or halting infection.
Decontaminating infant formula with the bacteriophage
When dealing with bacteria, antibiotics are usually the frontrunner, but there are cases where antibiotics are a big no. Take baby formula for instance, we cannot use antibiotics to keep bacteria at bay. This has posed a safety problem in recent years, but researchers have shown that we can use a natural enemy of bacteria to fight back without risk to infants’ health.
The science behind real life zombies

In the spirit of Halloween we bring you the science fact and fiction behind the undead. Zombies, those brain loving little guys, (and girls) are everywhere. We are all familiar (if you are horror fans, or at least not living on an Amish compound) with the classic zombie. But did you know that we aren’t the only zombie lovers out there? It turns out that nature has its own special types of zombies, but this isn’t a science fiction movie, this is science fact!
It’s alive!! Study adds to evidence that viruses are alive
The diverse physical attributes, genome sizes and lifestyles of viruses make them difficult to classify. A new study uses protein folds as evidence that viruses are living entities that belong on their own branch of the tree of life.
Image credit goes to: Julie McMahon
Classifying something as living isn’t as easy as it sounds, after all we are all atoms, so when do atoms go from nonliving to living? Despite the complexities of viruses, we have historically deemed them nonliving. However, a new analysis supports the hypothesis that viruses are living entities that share a long evolutionary history with cells. The study offers the first reliable method for tracing viral evolution back to a time when neither viruses nor cells existed in the forms recognized today.
An antibody that can attack HIV in new ways
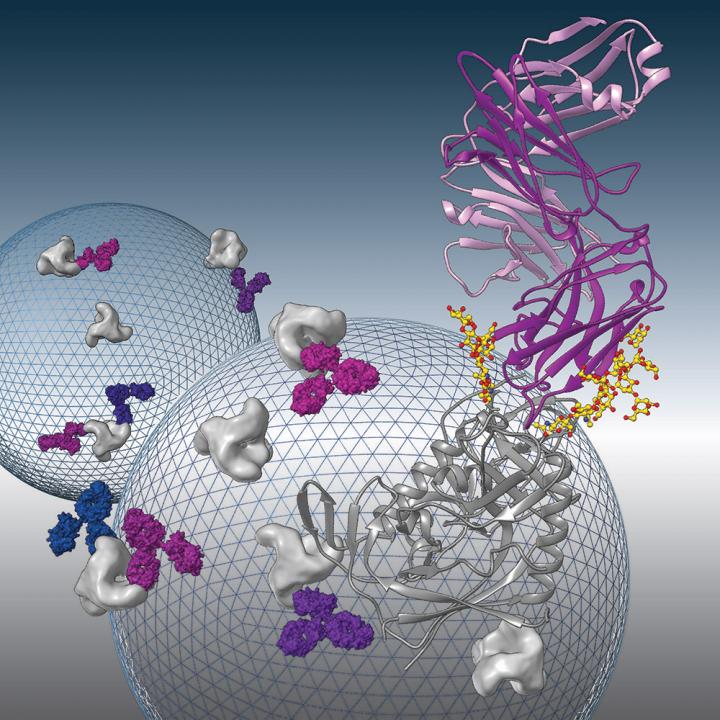
Broadly neutralizing antibodies to HIV-1 envelope glycoprotein are being evaluated as therapeutics to prevent or treat HIV-1 infection. Structural analysis of one such antibody, 8ANC195, revealed a new conformation of the envelope protein. The image shows the X-ray crystal structure of 8ANC195 in complex with the gp120 subunit of the envelope protein. The background shows schematic representations of HIV-1 virus particles studded with envelope proteins being recognized by 8ANC195 antibodies.
Image credit goes to: Louise Scharf/Caltech
Proteins called broadly neutralizing antibodies (bNAbs) are a promising key to the prevention of infection by HIV, the virus that causes AIDS. bNAbs have been found in blood samples from some HIV patients whose immune systems can naturally control the infection. These antibodies may protect a patient’s healthy cells by recognizing a protein called the envelope spike, present on the surface of all HIV strains and inhibiting, or neutralizing, the effects of the virus. Now Caltech researchers have discovered that one particular bNAb may be able to recognize this signature protein, even as it takes on different conformations during infection–making it easier to detect and neutralize the viruses in an infected patient.
HIV particles do not cause AIDS, our own immune cells do
Researchers have revealed that HIV does not cause AIDS by the virus’s direct effect on the host’s immune cells, but rather through the cells’ lethal influence on one another. HIV can either be spread through free-floating virus that directly infect the host immune cells or an infected cell can pass the virus to an uninfected cell.
How long have primates been infected with viruses related to HIV?
Disease-causing viruses engage their hosts in ongoing arms races: positive selection for antiviral genes increases host fitness and survival, and viruses in turn select for mutations that counteract the antiviral host factors. Studying such adaptive mutations can provide insights into the distant history of host-virus interactions. A study of antiviral gene sequences in African monkeys suggests that lentiviruses closely related to HIV have infected primates in Africa as far back as 16 million years.
New approach for making vaccines for deadly diseases
Researchers have devised an entirely new approach to vaccines – creating immunity without vaccination. The team has demonstrated that animals injected with synthetic DNA engineered to encode a specific neutralizing antibody against the dengue virus were capable of producing the exact antibodies necessary to protect against disease, without the need for standard antigen-based vaccination. Importantly, this approach, termed DMAb, was rapid, protecting animals within a week of administration.
Some vaccines support evolution of more-virulent viruses

This image shows chickens in agricultural production. Image credit goes to: Andrew Read, Penn State University
Scientific experiments with the herpes virus such as the one that causes Marek’s disease in poultry have confirmed, for the first time, the highly controversial theory that some vaccines could allow more-virulent versions of a virus to survive, putting unvaccinated individuals at greater risk of severe illness. The research has important implications for food-chain security and food-chain economics, as well as for other diseases that affect humans and agricultural animals.
Discovery points to a new path toward a universal flu vaccine
Flu vaccines can be something of a shot in the dark. Not only must they be given yearly, there’s no guarantee the strains against which they protect will be the ones circulating once the season arrives. New research by Rockefeller University scientists suggests it may be possible to harness a previously unknown mechanism within the immune system to create more effective and efficient vaccines against this ever-mutating virus.
A single mutation helped last year’s flu virus gain an advantage over the vaccine
The 2014-2015 flu vaccine didn’t work as well compared to previous years because the H3N2 virus recently acquired a mutation that concealed the infection from the immune system. A new study reveals the major viral mutation responsible for the mismatch between the vaccine strain and circulating strains. The research will help guide the selection of viral strains for future seasonal flu vaccines.
Hiding cells to prevent HIV transmission
The fight against HIV is ongoing and despite our rapid progression against the disease we still lack a cure or even adequate treatment for people infected. However, new research suggests that cloaking immune cells with antibodies that block T cell trafficking to the gut can substantially reduce the risk of viral transmission, at least in a non-human primate model of HIV infection. If it works out, this could help slow down the spread of HIV and give people a better shot at a normal life.
Scientists resurrect 700-year-old viruses, Just in time for Halloween!

You know how some zombie movies start with a discovery of a virus, it gets loose, and things quickly spiral out of control from that? Well in breaking news a team of researchers have found two 700-year-old viral sequences in frozen caribou dung in an arctic ice patch. The group isolated part of a viral RNA genome and the complete genome of a DNA virus. Then they infected living plants with the DNA virus, what could go wrong?